Advertisements
Advertisements
प्रश्न
In the electrolysis of aqueous sodium chloride solution which of the half cell reaction will occur at anode?
विकल्प
\[\ce{Na+ (aq) + e- -> Na (s); E^{Θ}_{cell} = - 2.71V}\]
\[\ce{2H2O (l) -> O2(g) + 4H+ (aq) + 4e^- ; E^{Θ}_{cell} = 1.23V}\]
\[\ce{H^+ (aq) + e^- -> 1/2 H2 (g); E^{Θ}_{cell} = 0.00V}\]
\[\ce{Cl^- (aq) -> 1/2 Cl2 (g) + e^- ; E^{Θ}_{cell} = 1.36V}\]
उत्तर
\[\ce{Cl^- (aq) -> 1/2 Cl2 (g) + e^- ; E^{Θ}_{cell} = 1.36V}\]
Explanation:
During electrolysis of aqueous
\[\ce{NaCl -> Na^+ + Cl^-}\]
\[\ce{H2O -> H+ + OH-}\]
\[\ce{Na+ + e- -> Na (E^{Θ}_{cell} = - 2.71V)}\]
\[\ce{H^+ e- -> 1/2 H2 E^{Θ}_{cell} = 0.00V}\]
At cathode,
\[\ce{H2O + e- -> 1/2 H2 + OH-}\]
At anode, two reactions are possible.
\[\ce{Cl^{-} -> 1/2 Cl2 + e- ; E^{Θ}_{cell} = 1.36V}\]
\[\ce{2H2O -> O2 + 4H+ + 4e- ; E^{Θ}_{cell} = 1.23V}\]
APPEARS IN
संबंधित प्रश्न
Write any four applications of electrochemical series
How much charge is required for the following reduction:
1 mol of \[\ce{Al^{3+}}\] to \[\ce{Al}\]?
How much charge is required for the following reduction:
1 mol of \[\ce{MnO^-_4}\] to \[\ce{Mn^{2+}}\]?
Write the name of the cell which is generally used in transistors. Write the reactions taking place at the anode and the cathode of this cell.
Following reactions occur at cathode during the electrolysis of aqueous copper(II) chloride solution :
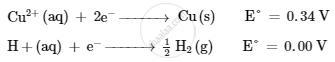
On the basis of their standard reduction electrode potential (E°) values, which reaction is feasible at the cathode and why ?
Assertion: Electrolysis of NaCl solution gives chlorine at anode instead of O2.
Reason: Formation of oxygen at anode requires overvoltage.
Given `1/a` = 0.5 CM–1, R = 50 ohm, N = 1.0 then equivalent conductance of electrolytic cell is
On Electrolysis of dilute sulphuric acid using platinum electrodes, the product obtained at the anode will be.
The quantity of electricity needed to separately electrolyse 1 M solution of ZnSO4, AlCl3, and AgNO3 completely is in the ratio of ______.
A current of 4 amp was passed for 2 hours through a solution of copper sulphate when 5.0 g of copper was deposited. The current efficiency is ______% (Cu = 63.5).
