Advertisements
Advertisements
प्रश्न
How much charge is required for the following reduction:
1 mol of \[\ce{MnO^-_4}\] to \[\ce{Mn^{2+}}\]?
उत्तर
\[\ce{\underset{(1 mol)}{MnO^-_4} + 8H^+ + \underset{(5 mol)}{5e^-} -> Mn^{2+} + 4H2O}\]
∴ Required charge = 5 Faradays
= 5 × 96500 C
= 4.825 × 105 C
APPEARS IN
संबंधित प्रश्न
On calculating the strength of current in amperes if a charge of 840C (coulomb) passes through an electrolyte in 7 minutes, it will be
- 1
- 2
- 3
- 4
Consider the reaction: \[\ce{Cr2O^{2-}_7 + 14H^+ + 6e^- -> 2Cr^{3+} + 7H2O}\]
What is the quantity of electricity in coulombs needed to reduce 1 mol of \[\ce{Cr2O^{2-}_7}\]?
A solution of \[\ce{Ni(NO3)2}\] is electrolysed between platinum electrodes using a current of 5 amperes for 20 minutes. What mass of \[\ce{Ni}\] is deposited at the cathode?
Draw neat labelled diagram of electrolytic refining of blister copper
Write the name of the cell which is generally used in transistors. Write the reactions taking place at the anode and the cathode of this cell.
Following reactions occur at cathode during the electrolysis of aqueous copper(II) chloride solution :
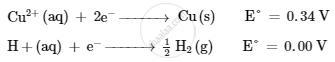
On the basis of their standard reduction electrode potential (E°) values, which reaction is feasible at the cathode and why ?
According to Faraday’s First Law of Electrolysis, the amount of chemical reaction which occurs at any electrode during electrolysis by a current is proportional to the ____________.
What will happen during the electrolysis of aqueous solution of \[\ce{CuSO4}\] by using platinum electrodes?
(i) Copper will deposit at cathode.
(ii) Copper will deposit at anode.
(iii) Oxygen will be released at anode.
(iv) Copper will dissolve at anode.
Assertion: Electrolysis of NaCl solution gives chlorine at anode instead of O2.
Reason: Formation of oxygen at anode requires overvoltage.
Consider the figure and answer the following question.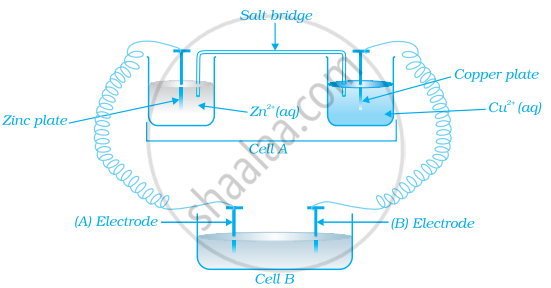
Cell ‘A’ has ECell = 2V and Cell ‘B’ has ECell = 1.1V which of the two cells ‘A’ or ‘B’ will act as an electrolytic cell. Which electrode reactions will occur in this cell?
Time Required to deposite one millimole of aluminium metal by the passage of 9.65 ampere through aqueous solution of aluminium is
When during electrolysis of a solution of Ag No3, 9650 coulombs of charge pass through the electroplating bath, the mass of silver deposite on the cathode will be:-
Given `1/a` = 0.5 CM–1, R = 50 ohm, N = 1.0 then equivalent conductance of electrolytic cell is
Through an aqueous solution of an unknown salt of metal M (M = 200 g/mol) a current of 1.93 A is passed for 50 min. If 4 g of metal is produced at cathode. The charge on metal ion in solution is ______.
On passing electricity through nitrobenzene solution, it is converted into azobenzene. The mass of azobenzene is ______ mg, if the same quantity of electricity produces oxygen just sufficient to burn 96 mg of fullerene (C60)·
Assertion (A): During electrolysis of aqueous copper sulphate solution using copper electrodes hydrogen gas is released at the cathode.
Reason (R): The electrode potential of Cu2+/Cu is greater than that of H+/H2.
Select the most appropriate answer from the options given below:
