Advertisements
Advertisements
प्रश्न
A solution of \[\ce{Ni(NO3)2}\] is electrolysed between platinum electrodes using a current of 5 amperes for 20 minutes. What mass of \[\ce{Ni}\] is deposited at the cathode?
उत्तर
The reaction takes place in the following manner-
\[\ce{Ni^{2+} + 2e^- -> Ni}\]
Atomic weight of \[\ce{Ni}\] = 58.70
Equivalent weight of \[\ce{Ni}\] = `("atomic weight")/("number of valence electrons")`
= `58.70/2`
= 29.35
According to Faraday's first law of electrolysis,
W = `"Z"."I"."t" = "Equivalent weight"/96500 xx "I" xx "t"`
= `29.35/96500 xx 5 xx 20 xx 60`
= 1.825 g
APPEARS IN
संबंधित प्रश्न
The charge of how many coulomb is required to deposit 1.0 g of sodium metal (molar mass 23.0 g mol-1) from sodium ions is -
- 2098
- 96500
- 193000
- 4196
How much electricity in terms of Faraday is required to produce 20 g of \[\ce{Ca}\] from molten \[\ce{CaCl2}\]?
(Given: Molar mass of Calcium is 40 g mol−1.)
State the first law of electrolysis
Following reactions occur at cathode during the electrolysis of aqueous sodium chloride solution:
Na+(aq) + e− ⟶ Na (s) E0 = 2.71 V
H+(aq) + e− ⟶ `1/2` H2 (g) E0 = 0.00 V
On the basis of their standard reduction electrode potential (E0) values, which reaction is feasible at the cathode and why?
How much charge is required for the following reduction:
1 mol of \[\ce{Cu^{2+}}\] to \[\ce{Cu}\]?
On passing 1.5 F charge, the number of moles of aluminium deposited at cathode are _______ [Molar mass of Al = 27 gram mol–1]
(A) 1.0
(B) 13.5
(C) 0.50
(D) 0.75
State second law of electrolysis
Explain Faraday’s second law of electrolysis
How many faradays of electricity are required to produce 13 gram of aluminium from aluminium chloride solution? (Given: Molar mass of Al = 27.0-gram mol–1)
Following reactions occur at cathode during the electrolysis of aqueous copper(II) chloride solution :
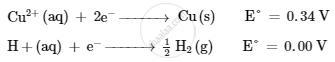
On the basis of their standard reduction electrode potential (E°) values, which reaction is feasible at the cathode and why ?
How much quantity of electricity in coulomb is required to deposit 1.346 × 10-3 kg of Ag in 3.5 minutes from AgNO3 solution?
( Given: Molar mass of Ag is 108 × 10-3 kg mol-1 )
Electrolytic cell uses electrical energy to bring about ____________.
Assertion: Electrolysis of NaCl solution gives chlorine at anode instead of O2.
Reason: Formation of oxygen at anode requires overvoltage.
Consider the figure and answer the following question.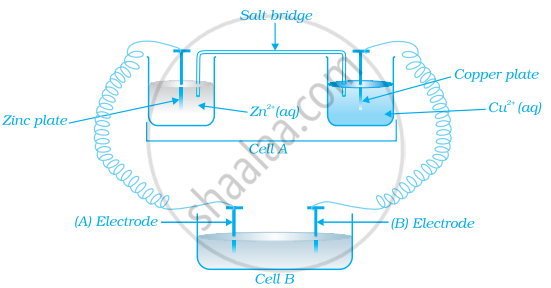
Cell ‘A’ has ECell = 2V and Cell ‘B’ has ECell = 1.1V which of the two cells ‘A’ or ‘B’ will act as an electrolytic cell. Which electrode reactions will occur in this cell?
Time Required to deposite one millimole of aluminium metal by the passage of 9.65 ampere through aqueous solution of aluminium is
When during electrolysis of a solution of Ag No3, 9650 coulombs of charge pass through the electroplating bath, the mass of silver deposite on the cathode will be:-
Given `1/a` = 0.5 CM–1, R = 50 ohm, N = 1.0 then equivalent conductance of electrolytic cell is
On Electrolysis of dilute sulphuric acid using platinum electrodes, the product obtained at the anode will be.
The quantity of electricity needed to separately electrolyse 1 M solution of ZnSO4, AlCl3, and AgNO3 completely is in the ratio of ______.
Assertion (A): During electrolysis of aqueous copper sulphate solution using copper electrodes hydrogen gas is released at the cathode.
Reason (R): The electrode potential of Cu2+/Cu is greater than that of H+/H2.
Select the most appropriate answer from the options given below:
