Advertisements
Advertisements
प्रश्न
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^23_11Na}\]
उत्तर
Geometric atomic structure of:
\[\ce{^23_11Na}\]
Number of protons p = 11 = e
A = p + n
23 = 11 + n
∴ Number of neutrons n = 12
Electronic configuration 2, 8, 1
K L M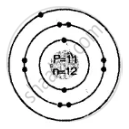
APPEARS IN
संबंधित प्रश्न
Mention the basic tendency of an atom which makes it to combine with other atoms.
Five atoms are labelled from A to E.
| Atoms | Mass No. | Atomic No. |
| A | 40 | 20 |
| B | 19 | 9 |
| C | 7 | 3 |
| D | 16 | 8 |
| E | 14 | 7 |
(a) Which one of these atoms:
(i) contains 7 protons,
(ii) has an electronic configuration 2, 7?
(b) Write down the formula of the compound formed between C and D.
(c) predict which are: (i) metals, (ii) non-metals?
What type of bond is formed between two atoms, when the electronegative difference between them is zero?
Name the following:
Metal with valency 2
Name the following:
Two atoms having the same number of protons and electrons but different number of neutrons.
Element M forms a chloride with the formula MCl2 which is solid with a high melting point. What type of bond is in MCl2. Write the formula of the compound when M combines with sulphur, oxygen, and nitrogen.
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^31_15P}\]
The element that would form cation due to the loss of electrons during the chemical reaction is ______.
______ theory explains the formation of molecules.
Choose the odd one out and write the reason:
