Advertisements
Advertisements
प्रश्न
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^12_6C}\]
उत्तर
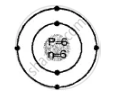
Geometric atomic structure of:
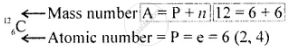
K shell = 2
L shell = 4
Electronic configuration [2, 4]
Electronic configuration [2, 4]
APPEARS IN
संबंधित प्रश्न
Give electron dot diagram of the following:
nitrogen
The following table shows the electronic configuration of the elements W, X, Y, Z:
|
Element |
W |
X |
Y |
Z |
|
Electronic |
2, 8, 1 |
2, 8, 7 |
2, 5 |
1 |
Answer the following question based on the table above:
What is the formula of the compound formed between W and X.
Name the following:
The shell closest to the nucleus of an atom
Choose the correct option
Which of the following is the correct electronic configuration of potassium?
State the type of bonding in the following molecules.
Water,
Identify the following reaction as either oxidation or reduction:
Zn → Zn2+ + 2e-
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^40_20Ca}\]
How do atoms attain Noble gas electronic configuration?
Complete the following table:
| Atoms | Atomic number | Electron distribution | |
| 1. | O | 8 | ______ |
| 2. | N | ______ | ______ |
| 3. | ______ | 17 | ______ |
| 4. | ______ | ______ | 2,8,2 |
How many electrons are required or released by each atom in chlorine to attain the nearest noble gas configuration?
