Advertisements
Advertisements
Question
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^12_6C}\]
Solution
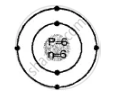
Geometric atomic structure of:
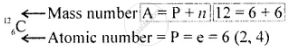
K shell = 2
L shell = 4
Electronic configuration [2, 4]
Electronic configuration [2, 4]
APPEARS IN
RELATED QUESTIONS
Match the atomic number 4, 8, 14, 15 and 19 with each of the following:
(a) A solid non-metal of valelncy 3.
(b) A gas of valency 2.
(c) A metal of valency 1.
(d) A non-metal of valency 4
Explain the following briefly:
Helium does not form He2 molecule
What type of bond is formed between two atoms, when the electronegative difference between them is High ?
Fill in the blanks.
The electrovalent bond or ionic bond is called as _____ bond
Give a reason
Physical properties of isotopes are different.
Explain
Duplet rule for the formation of hydrogen
Potassium (at No.19) and chlorine (at No.17) react to form a compound. Explain the formation of the compound on the basis of oxidation.
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^23_11Na}\]
What is a chemical bond?
How many atoms of each kind are present in the following molecule:
carbon tetrachloride
