Advertisements
Advertisements
प्रश्न
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^12_6C}\]
उत्तर
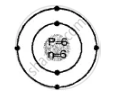
Geometric atomic structure of:
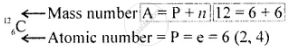
K shell = 2
L shell = 4
Electronic configuration [2, 4]
Electronic configuration [2, 4]
APPEARS IN
संबंधित प्रश्न
Elements A, B and C have atomic number 17, 19 and 10 respectively.
State which one is a non-metal.
Complete the following:
| Sodium | Phosphorus | Carbon | |
| Formula of chloride | |||
| Nature of bonding | |||
| Physical state of chloride |
Name the following:
An element having valency 'zero'
Give a reason
Physical properties of isotopes are different.
Give reason
`""_17^35"CI"` and `""_17^37"CI"` do not differ in their chemical reactions.
How do atoms attain noble gas configurations?
Give the orbital diagram of the following:
Methane
Identify the following reaction as either oxidation or reduction:
Fe3+ + e- → Fe2+
Explain Octet rule with an example.
Why are Noble gases inert in nature?
