Advertisements
Advertisements
प्रश्न
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^40_20Ca}\]
उत्तर
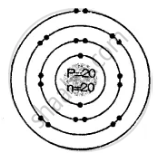
Geometric atomic structure of:
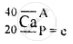
A = P + n
40 = 20 + n
∴ n = 40 − 20 = 20 neutrons
e = 20
Electronic arrangement 2, 8, 8, 2
K L M N
APPEARS IN
संबंधित प्रश्न
The following table shows the electronic configuration of the elements W, X, Y, Z:
|
Element |
W |
X |
Y |
Z |
|
Electronic |
2,8,1 |
2,8,7 |
2,5 |
1 |
Answer the following questions based on the table above:
What is the formula of the compound formed between X and Z.
How many atoms of each kind are present in the following molecules: calcium oxide, chlorine, water, carbon tetrachloride?
How many electrons are required by each atom mentioned in (a) to attain the nearest noble gas configuration?
An element M burns in oxygen to form an ionic compound MO. Write the formula of the compounds formed if this element is made to continue with chlorine and sulfur separately.
Give the orbital diagram of the following:
Methane
State the type of bonding in the following molecules.
Calcium oxide
Identify the following reaction as either oxidation or reduction:
O + 2e- → O2-
Write the basic concept of Kossel – Lewis theory.
How many electrons are required or released by each atom in chlorine to attain the nearest noble gas configuration?
Elements A, B and C have atomic number 17, 19 and 10 respectively.
State which one is chemically inert.
Elements A, B and C have atomic number 17, 19 and 10 respectively.
Write down the formula of the compound formed by two of the above elements.
