Advertisements
Advertisements
प्रश्न
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^40_20Ca}\]
उत्तर
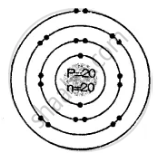
Geometric atomic structure of:
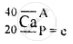
A = P + n
40 = 20 + n
∴ n = 40 − 20 = 20 neutrons
e = 20
Electronic arrangement 2, 8, 8, 2
K L M N
APPEARS IN
संबंधित प्रश्न
(i) How many atoms of each kind are present in the following molecules: calcium oxide, chlorine, water, carbon tetrachloride?
(ii) How many electrons are required for their octet structure?
Complete the following:
| Sodium | Phosphorus | Carbon | |
| Formula of chloride | |||
| Nature of bonding | |||
| Physical state of chloride |
An element with the atomic number 19 will most likely combine chemically with the element whose atomic number is
Why do `""_17^35"CI"` and `""_17^37"CI"` have the same chemical properties? In what respect do these atoms differ?
Give the orbital diagram of the following:
Nitrogen
Identify the following reaction as either oxidation or reduction:
Fe3+ + e- → Fe2+
In the formation of magnesium chloride (by direct combination between magnesium and chlorine), name the substance that is oxidized and the substance that is reduced.
The element that would form anion by gaining electrons in a chemical reaction is ______.
Elements with stable electronic configurations have eight electrons in their valence shell. They are ______.
The valency of noble gases is ______.
