Advertisements
Advertisements
प्रश्न
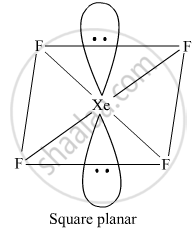
Draw the structure of XeF4.
उत्तर
XeF4
It has a square planar structure. Six electron pairs form an octahedron with two positions occupied by lone pairs.
संबंधित प्रश्न
Which noble gas has the lowest boiling point?
Which noble gas is used in filling balloons for meteorological observations?
Write the structures of the following molecules: XeOF4
Complete the following equation:
XeF2 + H2O →
What inspired N. Bartlett for carrying out reaction between Xe and PtF6?
How are XeO3 and XeOF4 prepared?
Write the electronic configuration of the following element:
Krypton (Z = 36)
Match the compounds given in Column I with the hybridisation and shape given in Column II and mark the correct option.
| Column I | Column II |
| (A) XeF6 | (1) sp3d3 – distorted octahedral |
| (B) XeO3 | (2) sp3d2 – square planar |
| (C) XeOF4 | (3) sp3 – pyramidal |
| (D) XeF4 | (4) sp3d2 – square pyramidal |
The order of increasing sizes of atomic radii among the elements O, S, Se and As is:
Noble gases are named because of their inertness towards reactivity. Identify an incorrect statement about them.
