Advertisements
Advertisements
Question
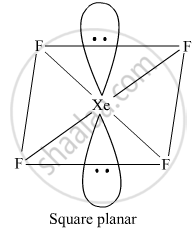
Draw the structure of XeF4.
Solution
XeF4
It has a square planar structure. Six electron pairs form an octahedron with two positions occupied by lone pairs.
RELATED QUESTIONS
Which noble gas is used in filling balloons for meteorological observations?
Write the structures of the following molecules: XeOF4
Draw the structures of the following:
XeO3
Balance the following equation: XeF6 + H2O → XeO2F2 + HF
Answer the following.
List the uses of Neon and argon gases.
Draw the structures of the following
XeF6
The number of lone pairs of electrons present in ClF5:
Partial hydrolysis of XeF4 gives ____________.
Match the compounds given in Column I with the hybridisation and shape given in Column II and mark the correct option.
| Column I | Column II |
| (A) XeF6 | (1) sp3d3 – distorted octahedral |
| (B) XeO3 | (2) sp3d2 – square planar |
| (C) XeOF4 | (3) sp3 – pyramidal |
| (D) XeF4 | (4) sp3d2 – square pyramidal |
\[\ce{XeF4 + H2O - \underline{}\underline{}\underline{}\underline{} + \underline{}\underline{}\underline{}\underline{}}\]
