Advertisements
Advertisements
प्रश्न
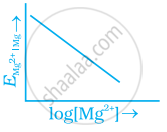
Electrode potential for Mg electrode varies according to the equation
`E_(Mg^(2+) | Mg) = E_(Mg^(2+) | Mg)^Θ - 0.059/2 log 1/([Mg^(2+)])`. The graph of `E_(Mg^(2+) | Mg)` vs `log [Mg^(2+)]` is ______.
विकल्प
उत्तर
Electrode potential for Mg electrode varies according to the equation
`E_(Mg^(2+) | Mg) = E_(Mg^(2+) | Mg)^Θ - 0.059/2 log 1/([Mg^(2+)])`. The graph of `E_(Mg^(2+) | Mg)` vs `log [Mg^(2+)]` is a straignt line with a positive slope and intercept `E_((Mg^(2+))/(Mg)`.

Explanation:
`E_(Mg^(2+) | mg) = E_(Mg^(2+) | Mg)^Θ - 0.059/2 log [Mg^(2+)]`
Compare this equation with the equation of straingt line y = mx + c.
The graph of `E_(Mg^(2+) | Mg)` versus `log [Mg^(2+)]` is a straignt line with a positive slope and intercept `E_((Mg^(2+))/(Mg)`.
APPEARS IN
संबंधित प्रश्न
A solution of CuSO4 is electrolysed using a current of 1.5 amperes for 10 minutes. What mass of Cu is deposited at cathode? [Atomic mass of Cu = 63.7]
If one mole electrons is passed through the solutions of CrCl3, AgNO3 and NiSO4, in what ratio Cr, Ag and Ni will be deposited at the electrodes?
Define anode
A copper electrode is dipped in 0.1 M copper sulphate solution at 25°C. Calculate the electrode potential of copper.
[Given: \[\ce{E^0_{{Cu^{2+}|Cu}}}\] = 0.34 V]
Consider a cell given below:
\[\ce{Cu | Cu^{2+} || Cl^{-} | Cl_{2},Pt}\]
Write the reactions that occur at anode and cathode
Which of the following statements about galvanic cell is incorrect
If the value of Ksp for Hg2Cl2 (s) is X then the value of X will be ____ where pX = - log X.
Given:
\[\ce{Hg2Cl2 + 2e- -> 2Hg(l) + 2Cl-}\], E° = 0.27 V
\[\ce{Hg+2 + 2e- -> 2Hg(l)}\] E° = 0.81 V
The two half cell reaction of an electrochemical cell is given as
\[\ce{Ag+ + e- -> Ag}\], `"E"_("Ag"^+//"Ag")^circ` = - 0.3995 V
\[\ce{Fe^{2+} -> Fe^{3+} + e-}\], `"E"_("Fe"^{3+}//"Fe")^{2+}` = - 0.7120 V
The value of EMF will be ______.
Galvanic cell is a device in which ______.
What should be the signs (positive/negative) for \[\ce{E^0_{cell}}\] and ΔG0 for a spontaneous redox reaction occurring under standard conditions?