Advertisements
Advertisements
Question
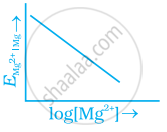
Electrode potential for Mg electrode varies according to the equation
`E_(Mg^(2+) | Mg) = E_(Mg^(2+) | Mg)^Θ - 0.059/2 log 1/([Mg^(2+)])`. The graph of `E_(Mg^(2+) | Mg)` vs `log [Mg^(2+)]` is ______.
Options
Solution
Electrode potential for Mg electrode varies according to the equation
`E_(Mg^(2+) | Mg) = E_(Mg^(2+) | Mg)^Θ - 0.059/2 log 1/([Mg^(2+)])`. The graph of `E_(Mg^(2+) | Mg)` vs `log [Mg^(2+)]` is a straignt line with a positive slope and intercept `E_((Mg^(2+))/(Mg)`.

Explanation:
`E_(Mg^(2+) | mg) = E_(Mg^(2+) | Mg)^Θ - 0.059/2 log [Mg^(2+)]`
Compare this equation with the equation of straingt line y = mx + c.
The graph of `E_(Mg^(2+) | Mg)` versus `log [Mg^(2+)]` is a straignt line with a positive slope and intercept `E_((Mg^(2+))/(Mg)`.
APPEARS IN
RELATED QUESTIONS
Why is anode in galvanic cell considered to be negative and cathode positive electrode?
Can Fe3+ oxidises bromide to bromine under standard conditions?
Given: \[\ce{E^0_{{Fe^{3+}|Fe^{2+}}}}\] = 0.771 V
\[\ce{E^0_{{Br_{2}|Br^-}}}\] = −1.09 V
Is it possible to store copper sulphate in an iron vessel for a long time?
Given: \[\ce{E^0_{{Cu^{2+}|{Cu}}}}\] = 0.34 V and \[\ce{E^0_{{Fe^{2+}|{Fe}}}}\] = −0.44 V
Two metals M1 and M2 have reduction potential values of −xV and +yV respectively. Which will liberate H2 and H2SO4.
Use the data given in below find out the most stable ion in its reduced form.
`"E"_("Cr"_2"O"_7^(2-)//"Cr"^(3+))^⊖`= 1.33 V `"E"_("Cl"_2//"Cl"^-)^⊖` = 1.36 V
`"E"_("MnO"_4^-//"Mn"^(2+))^⊖` = 1.51 V `"E"_("Cr"^(3+)//"Cr")^⊖` = - 0.74 V
For the given cell, \[\ce{Mg | Mg^{2+} || Cu^{2+} | Cu}\]
(i) \[\ce{Mg}\] is cathode
(ii) \[\ce{Cu}\] is cathode
(iii) The cell reaction is \[\ce{Mg^+ Cu^{2+} -> Mg^{2+} + Cu}\]
(iv) \[\ce{Cu}\] is the oxidising agent
Consider the following diagram in which an electrochemical cell is coupled to an electrolytic cell. What will be the polarity of electrodes ‘A’ and ‘B’ in the electrolytic cell?
If the half-cell reaction A + e– → A– has a large negative reduction potential, it follow that:-
The two half cell reaction of an electrochemical cell is given as
\[\ce{Ag+ + e- -> Ag}\], `"E"_("Ag"^+//"Ag")^circ` = - 0.3995 V
\[\ce{Fe^{2+} -> Fe^{3+} + e-}\], `"E"_("Fe"^{3+}//"Fe")^{2+}` = - 0.7120 V
The value of EMF will be ______.
Explain the types of electrochemical cells.