Advertisements
Advertisements
प्रश्न
Electrolysis is type of ______ reaction.
उत्तर
Electrolysis is the type of Decomposition reaction.
APPEARS IN
संबंधित प्रश्न
Taking into consideration the relationship in the first pair, complete the second pair
2H2 + O2 → 2H2O :Combination Reaction :: 2HgO → 2Hg + O2:_________
Give one example of a decomposition reaction which is carried out with electricity.
What type of chemical reaction is represented by the following equation?
X → Y + Z
What type of reaction is represented by the following equation?
NH4NO2 → N2 + 2H2O
MULTIPLE CHOICE QUESTIONS
Tick the most appropriate answer.
Thermal decomposition of a substance is brought about with the help of
- reactants
- water
- wind
- heat
Which one of the following processes involves chemical reactions?
Which of the following is an endothermic process?
When SO2 gas is passed through a saturated solution of H2S, which of the following reaction occurs?
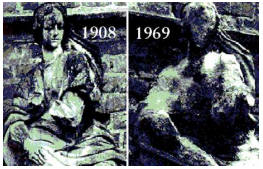
Marble statues are corroded or stained rain water. Identify the main reason.
On heating blue coloured powder of copper (II) nitrate, in a boiling tube, copper oxide (black), oxygen gas and a brown gas X is formed
- Write a balanced chemical equation of the reaction.
- Identify the brown gas X evolved.
- Identify the type of reaction.
- What could be the pH range of aqueous solution of the gas X?
