Advertisements
Advertisements
प्रश्न
Explain the following terms with suitable examples: F-centres
उत्तर
F-centres: When the anionic sites of a crystal are occupied by unpaired electrons, the ionic sites are called F-centres. These unpaired electrons impart colour to the crystals. For example, when crystals of NaCl are heated in an atmosphere of sodium vapour, the sodium atoms are deposited on the surface of the crystal. The Cl ions diffuse from the crystal to its surface and combine with Na atoms, forming NaCl. During this process, the Na atoms on the surface of the crystal lose electrons. These released electrons diffuse into the crystal and occupy the vacant anionic sites, creating F-centres.

APPEARS IN
संबंधित प्रश्न
If NaCl is doped with 10−3 mol % of SrCl2, what is the concentration of cation vacancies?
Yellow colour of NaCl crystals in sodium vapour is due to ____________.
The following diagram shows:
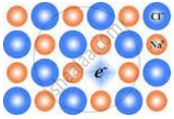
Which of the following will have metal deficiency defect?
Which of the following is a non-stoichiometric defect?
Pink colour of LiCl crystals is due to ______.
Why does table salt, NaCl, some times appear yellow in colour?
Why does white ZnO (s) becomes yellow upon heating?
Match the types of defect given in Column I with the statement given in Column II.
| Column I | Column II |
| (i) Impurity defect | (a) NaCl with anionic sites called F-centres |
| (ii) Metal excess defect | (b) FeO with Fe3+ |
| (iii) Metal deficiency defect | (c) NaCl with Sr2+ and some cationic sites vacant |
A sample of ferrous oxide has actual formula Fe0.93O1.00. In this sample what fraction of metal ions are Fe2+ ions? What type of nonstoichiometric defect is present in this sample?
The appearance of colour in solid alkali metal halides is generally due to
On doping Ge metal with a little of In (indium), we get:
The non-stoichiometric compound Fe0 940 is formed when x% of Fe2+ ions are replaced by as many `2/3` Fe3+ ions. The value of x is
If NaCl is doped with 10–3 mol % of SrCl2 calculate the concentration of cation vacancies.
Non-stoichiometric defects refer to ______
