Advertisements
Advertisements
प्रश्न
Explain the following terms with suitable examples : Interstitials
उत्तर
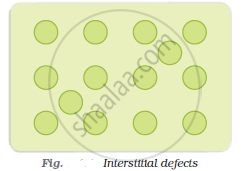
Interstitials: Interstitial defect is shown by non-ionic solids. This type of defect is created when some constituent particles (atoms or molecules) occupy an interstitial site of the crystal. The density of a substance increases because of this defect.
APPEARS IN
संबंधित प्रश्न
Give reasons : In stoichiometric defects, NaCl exhibits Schottky defect and not Frenkel defect.
Point defect is also known as ____________.
Dislocation defect is also known as ____________.
Type of stoichiometric defect shown by AgBr is ____________.
Assertion: No compound has both Schottky and Frenkel defects.
Reason: Both defects change the density of the solid.
Assertion: Due to Frenkel defect, there is no effect on the density of the crystalline solid.
Reason: In Frenkel defect, no cation or anion leaves the crystal.
Which of the following crystals does not exhibit Frenkel defect?
In a Schottky defect ____________.
Schottky defect is observed in crystals when ______.
Frenkel defect is also known as:
(i) stoichiometric defect
(ii) dislocation defect
(iii) impurity defect
(iv) non-stoichiometric defect
Why is FeO (s) not formed in stoichiometric composition?
Match the defects given in Column I with the statements in given Column II.
| Column I | Column II |
| (i) Simple vacancy defect | (a) shown by non-ionic solids and increases density of the solid. |
| (ii) Simple interstitial defect | (b) shown by ionic solids and decreases density of the solid. |
| (iii) Frenkel defect | (c) shown by non ionic solids and density of the solid decreases. |
| (iv) Schottky defect | (d) shown by ionic solids and density of the solid remains the same. |
Given below are two statements:
Statements I: Frenkel defects are vacancy as well as interstitial defects.
Statements II: Frenkel defect leads of colour in ionic solids due to the presence of F-centers.
Choose the most appropriate answer for the statements from the options given below:
The incorrect statement about the imperfections in solids is ______.
Which type of 'defect' has the presence of cations in the interstitial sites?
