Advertisements
Advertisements
प्रश्न
Schottky defect is observed in crystals when ______.
विकल्प
some cations move from their lattice site to interstitial sites.
equal number of cations and anions are missing from the lattice.
some lattice sites are occupied by electrons.
some impurity is present in the lattice.
उत्तर
Schottky defect is observed in crystals when equal number of cations and anions are missing from the lattice.
Explanation:
| Defect | Definition | Type of solid |
Effect of defect on density of substance |
Structure of crystal (with defect) |
| Schottky defect |
A crystal is said to have Schottky defect if equal number of cations and anions are missing from their normal lattice site there by creating vacancies or holes. |
Ionic solid |
Decreases | 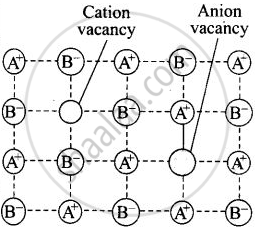 Schottky defect in a crystal |
APPEARS IN
संबंधित प्रश्न
Give reasons : In stoichiometric defects, NaCl exhibits Schottky defect and not Frenkel defect.
Frenkel defects are not found in alkali metal halides because ____________.
Type of stoichiometric defect shown by AgBr is ____________.
The following diagram shows:
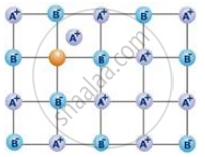
Schottky defect defines imperfection in the lattice structure of ____________.
Which of the following crystals does not exhibit Frenkel defect?
What is the effect of Frenkel defect on the density of ionic solids?
In a Schottky defect ____________.
Why is FeO (s) not formed in stoichiometric composition?
Schottky defect is noticed in ______
