Advertisements
Advertisements
प्रश्न
Why is FeO (s) not formed in stoichiometric composition?
उत्तर
In the crystals of FeO, some of the Fe2+ cations are replaced by Fe3+ ions. Three Fe2+ ions are replaced by two Fe3+ ions to make up for the loss of positive charge. Eventually there would be less amount of metal as compared to stoichiometric proportion.
APPEARS IN
संबंधित प्रश्न
Explain the following terms with suitable examples : Interstitials
What type of defect is shown by NaCl in
stoichiometric defects, and
Ionic solids containing large differences in sizes of ions show ____________.
Type of stoichiometric defect shown by ZnS is ____________.
The following diagram shows:
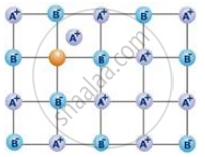
What is the effect of Frenkel defect on the density of ionic solids?
In a Schottky defect ____________.
Cations are present in the interstitial sites in ______.
Which of the following defects is also known as dislocation defect?
Which type of 'defect' has the presence of cations in the interstitial sites?
