Advertisements
Advertisements
प्रश्न
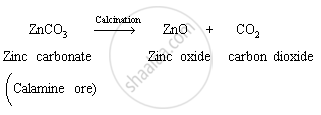
Explain given equation, what happens when ZnCO3 is heated in the absence of air?
उत्तर
When zinc carbonate is heated in the absence of air, it is converted to zinc oxide with the evolution of carbon dioxide gas. This is one of the methods used to convert concentrated metal carbonates to metal oxides during extraction of metals. This method is called calcination. The given zinc carbonate is nothing but calamine ore of zinc, which gets converted to zinc oxide on calcination.
APPEARS IN
संबंधित प्रश्न
What chemical process is used for obtaining a metal from its oxide?
Silver metal does not combine easily with oxygen but silver jewellery tarnishes after some time. How?
During galvanisation, iron metal is given a thin coating of one of the following metals. This metal is:
(a) chromium
(b) tin
(c) zinc
(d) copper
The articles made of silver metal become dark on prolonged exposure to air. This is due to the formation of a layer of its:
(a) oxide
(b) hydride
(c) sulphide
(d) carbonate
When an object made of metal A is kept in air for a considerable time, it loses its shine and becomes almost black due to the formation of a layer of substance B. When an object made of another metal C is kept in damp air for a considerable time, it gets covered with a green layer of substance D. Metal A is the best conductor of electricity whereas metal C is the next best conductor of electricity.
(a) What is metal A?
(b) What is metal C?
(c) Name the substance B.
(d) Name the substance D.
State three objectives achieved during the roasting of ores.
Aluminum is used in thermite welding:
write reaction for process?
The following question relate to the extraction of aluminium by electrolysis.
Give the equation for the reaction which takes place at the cathode.
How is the method of extraction of metals high up in the reactivity series different from that for metals in the middle? Why can the same process not be applied for them? Name the process used for the extraction of these metals.
Explain concept with example/explain with the help of a balanced equation.
Ionic bond and ionic compounds
