Advertisements
Advertisements
प्रश्न
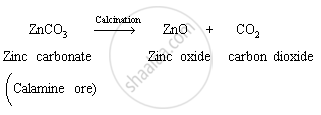
Explain given equation, what happens when ZnCO3 is heated in the absence of air?
उत्तर
When zinc carbonate is heated in the absence of air, it is converted to zinc oxide with the evolution of carbon dioxide gas. This is one of the methods used to convert concentrated metal carbonates to metal oxides during extraction of metals. This method is called calcination. The given zinc carbonate is nothing but calamine ore of zinc, which gets converted to zinc oxide on calcination.
APPEARS IN
संबंधित प्रश्न
Define the term ore.
The two metals which can be extracted just by heating their sulphides in air are:
(a) sodium and copper
(b) copper and aluminium
(c) potassium and zinc
(d) mercury and copper
Rock salt is an ore of one of the following metals. This metal is:
(a) Mn
(b) Na
(c) Fe
(d) Cu
The metal which can be extracted from pyrolusite ore is:
(a) mercury
(b) manganese
(c) aluminium
(d) magnesium
Zinc blende ore can be converted into zinc oxide by the process of:
(a) roasting
(b) hydrogenation
(c) chlorination
(d) calcination
Name the following:
Two metals which are found in the free or native state.
Froth floatation process is generally used to concentrate ______ ores.
Find the odd one out and give its explanation.
Which of the following ore is concentrated by the Froth floatation process?
Iqbal treated a lustrous, divalent element M with sodium hydroxide. He observed the formation of bubbles in reaction mixture. He made the same observations when this element was treated with hydrochloric acid. Suggest how can he identify the produced gas. Write chemical equations for both the reactions.
