Advertisements
Advertisements
Question
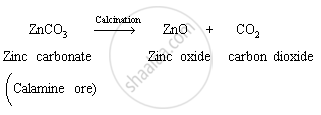
Explain given equation, what happens when ZnCO3 is heated in the absence of air?
Solution
When zinc carbonate is heated in the absence of air, it is converted to zinc oxide with the evolution of carbon dioxide gas. This is one of the methods used to convert concentrated metal carbonates to metal oxides during extraction of metals. This method is called calcination. The given zinc carbonate is nothing but calamine ore of zinc, which gets converted to zinc oxide on calcination.
APPEARS IN
RELATED QUESTIONS
How many carats is pure gold? Why is pure gold not suitable for making ornaments?
A zinc ore on heating in air forms sulphur dioxide. Describe briefly any two stages involved in the conversion of this concentrated ore into zinc metal.
What iron compound is present in haematite ore? Also write its chemical formula.
Name one ore of mercury. Which mercury compound is present in this ore? Write its chemical formula.
The major ore of aluminium is known as:
(a) cinnabar
(b) calamine
(c) bauxite
(d) pyrolusite
In an electrolytic tank, aluminium metal is being extracted by the electrolysis of molten aluminium oxide using carbon electrodes. It is observed that one of the carbon electrodes is gradually burnt away and has to be replaced.
(a) Which carbon electrode (cathode or anode) is burnt away?
(b) Why is this carbon electrode burnt away?
Which metal can be extracted from the following ore?
Calamine
An element has its electron configuration as 2,8,8,2. Now answer the following questions.
a) What is the atomic number of this element?
b) What is the group of this element?
c) To which period does this element belong?
Name the following:
The mixture of materials fed into a furnace to extract a metal.
Explain the following reaction with the balanced equation.
Chlorine dissolved in water
