Advertisements
Advertisements
प्रश्न
Explain how, a reduction reaction of aluminium can be used for welding cracked machine parts of iron. Write a chemical equation for the reaction involved.
उत्तर
When iron (III) oxide and aluminium powder are heated together, aluminium acts as a reducing agent and reduces iron (III) oxide to iron metal. It also gets oxidised to aluminium oxide with the liberation of excess heat. As excess heat is liberated in the reaction, the product, i.e., iron metal is formed in molten state. This reaction is an oxidation-reduction reaction and its chemical equation is:
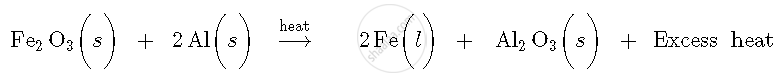
This reaction is called as thermite reaction. This reaction is widely used to weld cracked machine parts of iron. This is called thermite welding.
APPEARS IN
संबंधित प्रश्न
State one use of Aluminium.
Give the composition of an alloy called solder. State its one property and one use.
Which of the following alloys contains mercury as one of the constituents?
(a) stainless steel
(b) solder
(c) duralumin
(d) zinc amalgam
Mention two uses of the following metals and non-metals
Gold
Write ‘True’ or ‘False’ in front of following statement.
Magnesium is used in making light weight alloys of aluminium.
Match the alloys given in column I to the uses given in column II :
| Column I | Column II |
| (i) Duralumin | A. Electrical fuse |
| (ii) Solder | B. Surgical instruments |
| (iii) Brass | C. Aircraft body |
| (iv) Stainless Steel | D. Decorative articles |
Give reasons for the following:
Most metals conduct electricity well.
With reference to the following metals state their use in daily life metal:
copper
Compare the properties of metals and nonmetals. Give three examples for each.
The most abundant metal in the earth’s crust is ____________.
