Advertisements
Advertisements
Question
Explain how, a reduction reaction of aluminium can be used for welding cracked machine parts of iron. Write a chemical equation for the reaction involved.
Solution
When iron (III) oxide and aluminium powder are heated together, aluminium acts as a reducing agent and reduces iron (III) oxide to iron metal. It also gets oxidised to aluminium oxide with the liberation of excess heat. As excess heat is liberated in the reaction, the product, i.e., iron metal is formed in molten state. This reaction is an oxidation-reduction reaction and its chemical equation is:
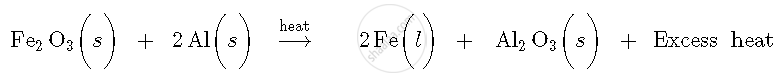
This reaction is called as thermite reaction. This reaction is widely used to weld cracked machine parts of iron. This is called thermite welding.
APPEARS IN
RELATED QUESTIONS
Give reasons for the following.
Sodium and potassium are stored in kerosene.
What happens when Dilute sulphuric acid is poured on a copper plate?
Write word equations of the reactions involved.
State one use of Silver.
Statements given below are incorrect. Write the correct statements :
Bronze is a mixture of 10% copper, 80% zinc and 10% tin.
Write name.
Molecular formula of the common ore of aluminium.
Which is the non-ionic compound in the compounds given below.
AgNO3 + Nacl → AgCl + NaNO3
What is it made from? Why?
Electric cables
With reference to the following metals state their use in daily life metal:
copper
Complete the following flow chart.

Why are bells made of metals?
