Advertisements
Advertisements
प्रश्न
Explain how, a reduction reaction of aluminium can be used for welding cracked machine parts of iron. Write a chemical equation for the reaction involved.
उत्तर
When iron (III) oxide and aluminium powder are heated together, aluminium acts as a reducing agent and reduces iron (III) oxide to iron metal. It also gets oxidised to aluminium oxide with the liberation of excess heat. As excess heat is liberated in the reaction, the product, i.e., iron metal is formed in molten state. This reaction is an oxidation-reduction reaction and its chemical equation is:
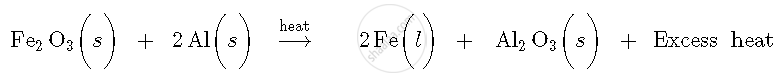
This reaction is called as thermite reaction. This reaction is widely used to weld cracked machine parts of iron. This is called thermite welding.
APPEARS IN
संबंधित प्रश्न
The atomic number of an element Y is 16.
(a) What must an atom of Y do to achieve the nearest inert gas electron arrangement?
(b) Which inert gas is nearest to Y?
Mention two uses of the following metals and non-metals
Gold
Mention two uses of the following metals and non-metals
Oxygen
Give the general characteristics of:
Alkaline earth metals with reference to
(i) bonding
(ii) action of air
(iii) action of water
(iv) action of acid
Take an element from an alkali metal and one from an alkaline earth metal and write an equation for their action with:
Hydrochloric acid,
State the position of the following in the periodic table:
Halogens
What is it made from? Why?
A pair of tongs.
Name the following:
A metal used in flexible pipes.
Which one of the following four metals would be displaced from the solution of its salts by other three metals?
The liquid metal at room temperature ______.
