Advertisements
Advertisements
प्रश्न
Explain the following
A glass bottle completely filled with water and tightly closed at room temperature is likely to burst when kept in the freezer of a refrigerator.
उत्तर
Inside the freezer, when the temperature of water falls below 4°C, the water in the bottle starts expanding. If the bottle is completely filled and tightly closed, there is no space for water to expand, and hence, the bottle may burst.
APPEARS IN
संबंधित प्रश्न
Give reason of Ice floats on water.
Observe the following graph. Considering the change in volume of water as its temperature is raised from 0°C, discuss the difference in the behaviour of water and other substances. What is this behaviour of water called?

A 50 kg man is running at a speed of 18 km h−1. If all the kinetic energy of the man can be used to increase the temperature of water from 20°C to 30°C, how much water can be heated with this energy?
Describe an experiment to show that water has maximum density at 4°C. What important consequences follow this peculiar property of water? Discuss the importance of this phenomenon in nature.
Explain the following
A hollow glass sphere which floats with its entire volume submerged in water at 4°C, sinks when water is heated above 4°C.
What do you understand by the anomalous expansion of water?
How do fishes survive in frozen lakes?
Explain, why are soft drink bottles not completely filled?
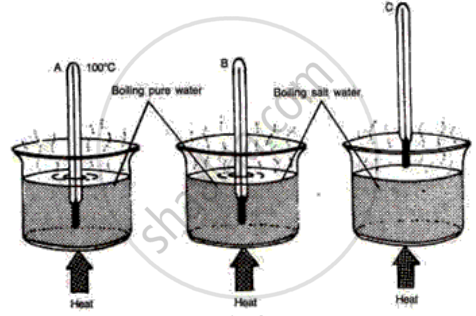
The following diagrams illustrate three situations involving thermometers which are labeled A, Band C. In each situation the thermometers indicate different readings.
(i) What do you expect the approximate reading of the thermometer B and C would be? Give a reason for your answer.
(ii) How would the readings of A and B help you in calibrating a thermometer?