Advertisements
Advertisements
प्रश्न
Explain the following reaction:
Cannizzaro reaction
उत्तर
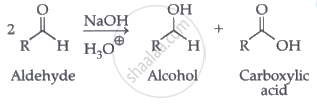
Aldehydes which do not have an α-hydrogen atom, undergo self oxidation and reduction (disproportionation) reaction on heating with concentrated alkali. In this reaction, one molecule of the aldehyde is reduced to alcohol while another is oxidised to carboxylic acid salt.
APPEARS IN
संबंधित प्रश्न
Write the chemical equations to illustrate the following name reaction:
Cannizzaro’s reaction
Describe the following:
Cannizzaro reaction
Write the reactions involved in the following reactions: Clemmensen reduction
Write the equations involved in the following reactions:
Etard reaction
Write the product formed when p-nitro chlorobenzene is heated with aqueous NaOH at 443K followed by acidification?
complete the following reaction:
\[\begin{array}{cc}
\phantom{...}\ce{CH3} \\
| \\
\phantom{.................}\ce{CH3-CH-COOH ->[(i) Br2/Red P4][(ii)H2O]}
\end{array}\]
Complete the following reaction:

The products obtained in the Cannizzaro reaction are
\[\begin{array}{cc}
\ce{D}\phantom{........................}\\
|\phantom{.........................}\\
\ce{2D - C = O + OH^- ->[Cannizzaro] X and Y}
\end{array}\]
(Y is alcohol, D is deuterium)
X and Y will have the structure:
Which of the following does not give Cannizzaro reaction?
