Advertisements
Advertisements
प्रश्न
Explain the following reactions:
Stephan reaction
उत्तर
This reaction is used to synthesize aldehydes from nitriles or cyanides. Firstly, nitriles or cyanides are reduced in presence of stannous chloride and hydrochloric acid in ethyl acetate solvent to form imine intermediate. Then hydrolysis of this intermediate with water gives the corresponding aldehyde.
\[\ce{CH3 - C ≡ N + 2[H] ->[SnCl2/HCl][Ether] CH3 - CH = NH + HCl ->[H2O/H^+] \underset{Acetaldehyde}{CH3CHO} + NH4Cl}\]
APPEARS IN
संबंधित प्रश्न
How will you bring about the following conversion?
Benzoyl chloride to benzaldehyde
Write the structure of the product of the following reaction:
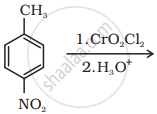
Aldehydes are produced on reduction of the following by DIBAL-H:
Aldehydes are prepared by reducing nitriles to corresponding imines with stannous chloride in the presence of hydrochloric acid. This reaction is called:
Name the electrophile produced in the reaction of benzene with benzoyl chloride in the presence of anhydrous \[\ce{AlCl3}\]. Name the reaction also.
Ethylbenzene is generally prepared by acetylation of benzene followed by reduction and not by direct alkylation. Think of a possible reason.
The general formula CnH2NO2 could be for open chain
The reagent in friedel - craft reaction is:
The oxidation of toluene to benzaldehyde by chromyl chloride is called ______.
An organic compound with molecular formula \[\ce{C7H7NO2}\] exists in three isomeric forms, the isomer ‘A’ has the highest melting point of the three. ‘A’ on reduction gives compound ‘B’ with molecular formula \[\ce{C7H9N}\]. ‘B’ on treatment with \[\ce{NaNO2/HCl}\] at 0-5° C to form compound ‘C’. On treating C with \[\ce{H3PO2}\], it gets converted to D with formula \[\ce{C7H8}\], which on further reaction with \[\ce{CrO2Cl2}\] followed by hydrolysis forms ‘E’ \[\ce{C7H6O}\]. Write the structure of compounds A to E. Write the chemical equations involved.
