Advertisements
Advertisements
प्रश्न
The oxidation of toluene to benzaldehyde by chromyl chloride is called ______.
विकल्प
Etard reaction
Riemer-Tiemann reaction
Stephen’s reaction
Cannizzaro’s reaction
उत्तर
The oxidation of toluene to benzaldehyde by chromyl chloride is called Etard reaction.
Explanation:
The oxidation of toluene (C6H5CH3) with chromyl chloride (CrO2Cl2) in CCl4 or CS2 to give benzaldehyde is called Etard reaction. In this reaction, the chromyl chloride first forms a brown complex. which is separated and then decomposed with H2O to give benzaldehyde (C6H5CHO).
APPEARS IN
संबंधित प्रश्न
Write the product in the following reaction:
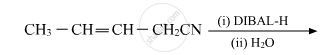
Write the chemical equations to illustrate the following name reactions : Rosenmund reduction
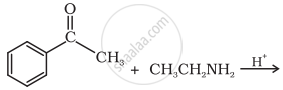
Predict the product of the following reaction:
The oxidation of toluene to benzoic acid can be done using which of the following reagents.
In the chromyl chloride test, the final step results in the formation of a yellow precipitate of the following:
When 2 – hydroxyl benzoic acid distilled with zinc dust, it give
Aldehydes are the first oxidation products of ______.
The intermediate compound ‘X’ in the following chemical reaction is:
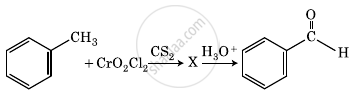
Convert the following:
Benzoic acid to Benzaldehyde
Account for the following:
Reduction of nitrobenzene using Fe and HCl is preferred over Sn and HCl.
