Advertisements
Advertisements
प्रश्न
The oxidation of toluene to benzaldehyde by chromyl chloride is called ______.
पर्याय
Etard reaction
Riemer-Tiemann reaction
Stephen’s reaction
Cannizzaro’s reaction
उत्तर
The oxidation of toluene to benzaldehyde by chromyl chloride is called Etard reaction.
Explanation:
The oxidation of toluene (C6H5CH3) with chromyl chloride (CrO2Cl2) in CCl4 or CS2 to give benzaldehyde is called Etard reaction. In this reaction, the chromyl chloride first forms a brown complex. which is separated and then decomposed with H2O to give benzaldehyde (C6H5CHO).
APPEARS IN
संबंधित प्रश्न
Write the product in the following reaction
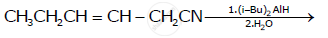
Ozonolysis of alkenes followed by the reaction with zinc dust and water gives ____________ depending on the substitution pattern of the alkene.
The oxidation of toluene to benzoic acid can be stopped at the aldehyde stage to give benzaldehyde. The reagent used for the purpose is one of the following.
The oxidation of toluene to benzoic acid can be done using which of the following reagents.
Benz aldehyde + NaOH →
The reagent in friedel - craft reaction is:
Explain the following reactions:
Stephan reaction
The reaction of benzene with CO and HCl in the presence of anhydrous AlCl3 gives ______.
Account for the following:
N-ethylbenzene sulphonyl amide is soluble in alkali.
Write the name of the reaction, structure and IUPAC name of the product formed when:
CH3CH2CN reacts with stannous chloride in the presence of hydrochloric acid, followed by hydrolysis.
