Advertisements
Advertisements
प्रश्न
Write the name of the reaction, structure and IUPAC name of the product formed when:
CH3CH2CN reacts with stannous chloride in the presence of hydrochloric acid, followed by hydrolysis.
उत्तर
Stephen reaction, CH3CH2CHO, Propanal.
APPEARS IN
संबंधित प्रश्न
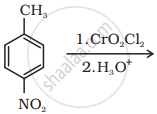
Write the structure of the product of the following reaction:
When 0.4g of oxalic acid is dissolved in 40g of benzene, the freezing point of the solution is lowered by 0.45 K. Calculate the degree of association of acetic acid. Acetic acid forms dimer when dissolved in benzene.
(Kf for benzene = 5.12 K kg mol-1, at. wt. C = 12, H = 1, O = 16)
The oxidation of toluene to benzoic acid can be done using which of the following reagents.
An aromatic compound ‘A’ (Molecular formula \[\ce{C8H8O}\]) gives positive 2, 4-DNP test. It gives a yellow precipitate of compound ‘B’ on treatment with iodine and sodium hydroxide solution. Compound ‘A’ does not give Tollen’s or Fehling’s test. On drastic oxidation with potassium permanganate it forms a carboxylic acid ‘C’ (Molecular formula \[\ce{C7H6O2}\]), which is also formed along with the yellow compound in the above reaction. Identify A, B and C and write all the reactions involved.
The general formula CnH2NO2 could be for open chain
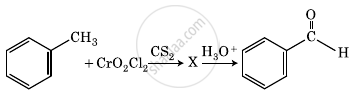
The intermediate compound ‘X’ in the following chemical reaction is:
Predict the reagent for carrying out the following transformations:
Benzoyl chloride to Benzaldehyde
The reaction of benzene with CO and HCl in the presence of anhydrous AlCl3 gives ______.
Reagent used to convert allyl alcohol to acrolein is ______.
Account for the following:
N-ethylbenzene sulphonyl amide is soluble in alkali.
