Advertisements
Advertisements
प्रश्न
Account for the following:
N-ethylbenzene sulphonyl amide is soluble in alkali.
उत्तर
The hydrogen attached to N-Ethylbenzene sulphonamide is acidic in nature. This is due to the presence of a strong electron-withdrawing sulphonyl group. Hence, it is soluble in alkali.
APPEARS IN
संबंधित प्रश्न
Write the product in the following reaction
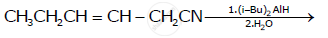
Write the chemical equations to illustrate the following name reactions : Rosenmund reduction
Aldehydes are produced on reduction of the following by DIBAL-H:
The oxidation of toluene to benzoic acid can be stopped at the aldehyde stage to give benzaldehyde. The reagent used for the purpose is one of the following.
What is the name of the given reaction of preparation of aldehyde?
\[\ce{C3COCl ->[H2][Pd/BaSO4] CH3CHO + HCl}\]
Benz aldehyde + NaOH →
When 2 – hydroxyl benzoic acid distilled with zinc dust, it give
Convert the following:
Benzoic acid to Benzaldehyde
Explain the following reactions:
Stephan reaction
Write the name of the reaction, structure and IUPAC name of the product formed when:
CH3CH2CN reacts with stannous chloride in the presence of hydrochloric acid, followed by hydrolysis.
