Advertisements
Advertisements
प्रश्न
Account for the following:
N-ethylbenzene sulphonyl amide is soluble in alkali.
उत्तर
The hydrogen attached to N-Ethylbenzene sulphonamide is acidic in nature. This is due to the presence of a strong electron-withdrawing sulphonyl group. Hence, it is soluble in alkali.
APPEARS IN
संबंधित प्रश्न
How will you bring about the following conversion?
Benzoyl chloride to benzaldehyde
Ozonolysis of alkenes followed by the reaction with zinc dust and water gives ____________ depending on the substitution pattern of the alkene.
The oxidation of toluene to benzoic acid can be stopped at the aldehyde stage to give benzaldehyde. The reagent used for the purpose is one of the following.
An alkene ‘A’ (Mol. formula \[\ce{C5H10}\]) on ozonolysis gives a mixture of two compounds ‘B’ and ‘C’. Compound ‘B’ gives positive Fehling’s test and also forms iodoform on treatment with \[\ce{I2}\] and \[\ce{NaOH}\]. Compound ‘C’ does not give Fehling’s test but forms iodoform. Identify the compounds A, B and C. Write the reaction for ozonolysis and formation of iodoform from B and C.
The number of chiral carbon in glucose is:-
The general formula CnH2NO2 could be for open chain
The intermediate compound ‘X’ in the following chemical reaction is:
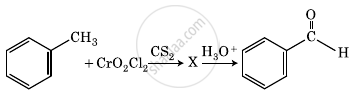
Convert the following:
Benzoic acid to Benzaldehyde
Predict the reagent for carrying out the following transformations:
Benzoyl chloride to Benzaldehyde
An organic compound with molecular formula \[\ce{C7H7NO2}\] exists in three isomeric forms, the isomer ‘A’ has the highest melting point of the three. ‘A’ on reduction gives compound ‘B’ with molecular formula \[\ce{C7H9N}\]. ‘B’ on treatment with \[\ce{NaNO2/HCl}\] at 0-5° C to form compound ‘C’. On treating C with \[\ce{H3PO2}\], it gets converted to D with formula \[\ce{C7H8}\], which on further reaction with \[\ce{CrO2Cl2}\] followed by hydrolysis forms ‘E’ \[\ce{C7H6O}\]. Write the structure of compounds A to E. Write the chemical equations involved.
