Advertisements
Advertisements
प्रश्न
Explain Why?
Inflating a balloon seems to violate Boyle's law.
उत्तर
Inflating a balloon seems violating Boyles law as volume is increasing with increase in pressure. Since the mass of gas is also increasing.
APPEARS IN
संबंधित प्रश्न
Explain Boyle's Law on the basis of the kinetic theory of matter.
State the law which is represented by the following graph:
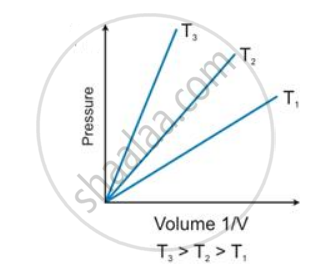
Give reasons for the following:
It is necessary to specify the pressure and temperature of gas while stating its volume.
Choose the correct answer:
If the pressure is doubled for a fixed mass of a gas, its volume will become
Calculate the volume occupied by 2 g of hydrogen at 27°C and 4-atmosphere pressure if at STP it occupies 22.4 litres.
50 cm3 of hydrogen is collected over water at 17°C and 750 mmHg pressure. Calculate the volume of a dry gas at STP. The water vapour pressure at 17°C is 14 mmHg.
Fill in the blank with the correct word, from the words in option:
If the pressure of a fixed mass of a gas is kept constant and the temperature is increased, the volume correspondingly _______
State Boyle’s law
State-the law of volume
The following question refers to one mole of chlorine gas.
What will happen to volume of gas, if pressure is doubled?
