Advertisements
Advertisements
प्रश्न
Fill in the blank with the correct word, from the words in option:
If the pressure of a fixed mass of a gas is kept constant and the temperature is increased, the volume correspondingly _______
विकल्प
increases
decreases
उत्तर
If the pressure of a fixed mass of a gas is kept constant and the temperature is increased, the volume correspondingly increases.
APPEARS IN
संबंधित प्रश्न
Explain Why?
Inflating a balloon seems to violate Boyle's law.
Explain Boyle's Law on the basis of the kinetic theory of matter.
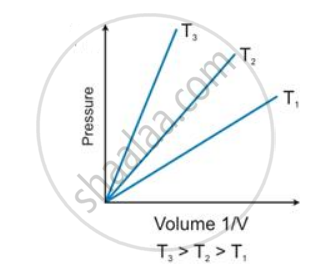
State the law which is represented by the following graph:
561 dm3 of a gas at STP is filled in a 748 dm3 container. If the temperature is constant, calculate the percentage change in pressure required.
A given mass of a gas occupied 143 cm3 at 27° C and 700 mm Hg pressure. What will be its volume at 300 K and 280 mm Hg pressure?
Calculate the following:
Calculate the temperature at which a gas ‘A’ at 20°C having a volume, of 500 cc. will occupy a volume of 250 cc.
Assuming temperature remaining constant calculate the pressure of the gas in the following:
The pressure of a gas having volume 1000 cc. originally occupying 1500 cc. at 720 mm. pressure.
Assuming temperature remaining constant calculate the pressure of the gas in the following:
The pressure of a gas having volume 1800 ml. originally occupying 300 ml. at 6 atms. pressure.
Calculate the coefficient of cubical expansion of a zinc bar. Whose volume is increased 0.25 m3 from 0.3 m3 due to the change in its temperature of 50K?
The following question refers to one mole of chlorine gas.
What will happen to volume of gas, if pressure is doubled?
