Advertisements
Advertisements
Question
Fill in the blank with the correct word, from the words in option:
If the pressure of a fixed mass of a gas is kept constant and the temperature is increased, the volume correspondingly _______
Options
increases
decreases
Solution
If the pressure of a fixed mass of a gas is kept constant and the temperature is increased, the volume correspondingly increases.
APPEARS IN
RELATED QUESTIONS
State the law which is represented by the following graph:
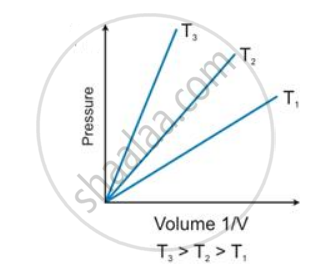
At 0°C and 760 mmHg pressure, a gas occupies a volume of 100 cm3. Kelvin temperature of the gas is increased by one-fifth and the pressure is increased one and a half times. Calculate the final volume of the gas.
A certain mass of a gas occupies 2 litres at 27°C and 100 Pa. Find the temperature when volume and pressure become half of their initial values.
22.4 litres of gas weighs 70 g at STP. Calculate the weight of the gas if it occupies a volume of 20 litres at 27°C and 700 mmHg of pressure.
A gas cylinder having a capacity of 20 litres contains a gas at 100 atmos. How many flasks of 200 cm3 capacity can be filled from it at 1 atmos. pressure if the temperature remains constant?
Name or state the following:
The law which studies the relationship between pressure of a gas and the volume occupied by it at constant temperature.
Calculate the volume of a gas ‘A’ at s.t.p., if at 37°C and 775 mm of mercury pressure, it occupies a volume of `9 1/2` litres.
Assuming temperature remaining constant calculate the pressure of the gas in the following:
The pressure of a gas having volume 1500 cm3 originally occupying 750 cc. at 5 ats. pressure.
Fill in the blank with the correct word, from the words in option:
1 dm3 of a gas is equal to _______.
According to Boyle’s law, the shape of the graph between pressure and reciprocal of volume is _______.
