Advertisements
Advertisements
Question
State the law which is represented by the following graph:
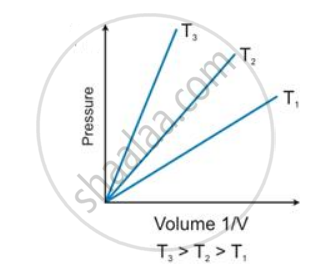
Solution
Boyle's law
APPEARS IN
RELATED QUESTIONS
Give reasons for the following:
It is necessary to specify the pressure and temperature of gas while stating its volume.
At constant temperature, the effect of change of pressure on the volume of a gas was as given below:
|
Pressure in atmosphere |
Volume in liters |
|
0.20 |
112 |
|
0.25 |
89.2 |
|
0.40 |
56.25 |
|
0.60 |
37.40 |
|
0.80 |
28.10 |
|
1.00 |
22.4 |
(a) Plot the following graphs
- P vs V
- P vs 1/V
- PV vs P
Interpret each graph in terms of the law.
(b) Assuming that the pressure values given above are correct, find the correct measurement of the volume.
A certain mass of a gas occupies 2 litres at 27°C and 100 Pa. Find the temperature when volume and pressure become half of their initial values.
A given mass of a gas occupied 143 cm3 at 27° C and 700 mm Hg pressure. What will be its volume at 300 K and 280 mm Hg pressure?
Assuming temperature remaining constant calculate the pressure of the gas in the following:
The pressure of a gas having volume 1000 cc. originally occupying 1500 cc. at 720 mm. pressure.
Assuming temperature remaining constant calculate the pressure of the gas in the following:
The pressure of a gas having volume 100 lits. originally occupying 75 dm3 at 700 mm. pressure.
Fill in the blank with the correct word, from the words in option:
If the pressure of a fixed mass of a gas is kept constant and the temperature is increased, the volume correspondingly _______
According to Boyle’s law, the shape of the graph between pressure and reciprocal of volume is _______.
State-the law of volume
Calculate the coefficient of cubical expansion of a zinc bar. Whose volume is increased 0.25 m3 from 0.3 m3 due to the change in its temperature of 50K?
