Advertisements
Advertisements
प्रश्न
State the law which is represented by the following graph:
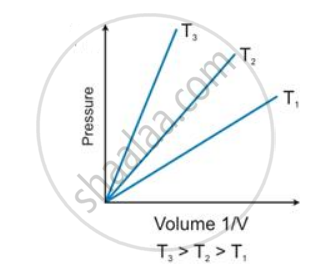
उत्तर
Boyle's law
APPEARS IN
संबंधित प्रश्न
Explain Boyle's Law on the basis of the kinetic theory of matter.
Choose the correct answer:
If the pressure is doubled for a fixed mass of a gas, its volume will become
561 dm3 of a gas at STP is filled in a 748 dm3 container. If the temperature is constant, calculate the percentage change in pressure required.
State Boyle's Law.
Correct the following statement:
The volume of a gas is inversely proportional to its pressure at a constant temperature.
Calculate the following:
A gas ‘X’ is collected over water at 17°C and 750 mm. pressure. If the volume of the gas collected is 50 cc., calculate the volume of the dry gas at s.t.p. [at 17°C the vapour pressure is 14 mm.]
Assuming temperature remaining constant calculate the pressure of the gas in the following:
The pressure of a gas having volume 1000 cc. originally occupying 1500 cc. at 720 mm. pressure.
According to Boyle’s law, the shape of the graph between pressure and reciprocal of volume is _______.
State Boyle’s law
State-the law of volume
