Advertisements
Advertisements
Question
A gas cylinder having a capacity of 20 litres contains a gas at 100 atmos. How many flasks of 200 cm3 capacity can be filled from it at 1 atmos. pressure if the temperature remains constant?
Solution
V1 = 20 lits. = 20000 cc
V2 = ?
P1 = 100 atm
P2 = 1 atm
At constant temperature
P1V1 = P2V2
`"V"_2 = ("P"_1"V"_1)/"P"_2 = (100 xx 20000)/1 = 2000000` cc
∴ Number of flasks of capacity 200 cc
n × 200 = 2000000
n = `2000000/200 = 10000` flasks
APPEARS IN
RELATED QUESTIONS
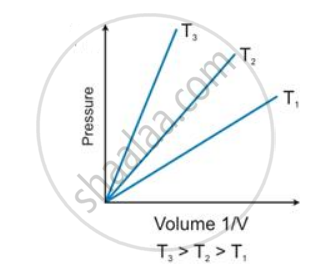
State the law which is represented by the following graph:
Give reasons for the following:
It is necessary to specify the pressure and temperature of gas while stating its volume.
561 dm3 of a gas at STP is filled in a 748 dm3 container. If the temperature is constant, calculate the percentage change in pressure required.
A certain amount of a gas occupies a volume of 0.4 litre at 17°C. To what temperature should it be heated so that its volume gets (a) doubled, (b) reduced to half, pressure remaining constant?
A certain mass of a gas occupies 2 litres at 27°C and 100 Pa. Find the temperature when volume and pressure become half of their initial values.
State Boyle's Law.
Calculate the following:
Calculate the temperature at which a gas ‘A’ at 20°C having a volume, of 500 cc. will occupy a volume of 250 cc.
Calculate the following:
A gas ‘X’ is collected over water at 17°C and 750 mm. pressure. If the volume of the gas collected is 50 cc., calculate the volume of the dry gas at s.t.p. [at 17°C the vapour pressure is 14 mm.]
According to Boyle’s law, the shape of the graph between pressure and reciprocal of volume is _______.
State-the law of volume
