Advertisements
Advertisements
Question
A certain amount of a gas occupies a volume of 0.4 litre at 17°C. To what temperature should it be heated so that its volume gets (a) doubled, (b) reduced to half, pressure remaining constant?
Solution
(a) V1 = 0.4 L
V2 = 0.4 × 2L
T1 = 17°C (17 + 273) = 290 K
T2= ?
`("V"_1)/("T"_1) = "V"_2/"T"_2`
`0.4/290 = 0.8/"T"_2`
T2 = 290 x 2 = 580
T2 = 580 - 273 = 307 °C
(b) V1 = 0.4 L
V2 = 0.2 L
T1 = 17°C (17 + 273) = 290 K
T2= ?
`"V"_1/"T"_1 = "V"_2/"T"_2`
`0.4/290 = 0.2/"T"_2`
T2 = `(290 xx 0.2)/0.4 = 145 "K"`
T2 = 145 - 273 = -128 °C
APPEARS IN
RELATED QUESTIONS
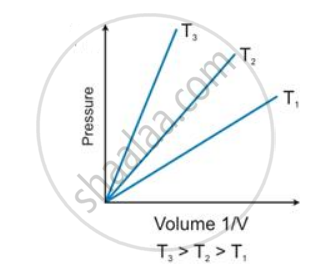
State the law which is represented by the following graph:
Calculate the volume occupied by 2 g of hydrogen at 27°C and 4-atmosphere pressure if at STP it occupies 22.4 litres.
Calculate the volume of dry air at STP that occupies 28 cm3 at 14°C and 750 mmHg pressure when saturated with water vapour. The vapour pressure of water at 14°C is 12 mmHg.
22.4 litres of gas weighs 70 g at STP. Calculate the weight of the gas if it occupies a volume of 20 litres at 27°C and 700 mmHg of pressure.
Correct the following statement:
The volume of a gas is inversely proportional to its pressure at a constant temperature.
Calculate the following:
A gas ‘X’ is collected over water at 17°C and 750 mm. pressure. If the volume of the gas collected is 50 cc., calculate the volume of the dry gas at s.t.p. [at 17°C the vapour pressure is 14 mm.]
Assuming temperature remaining constant calculate the pressure of the gas in the following:
The pressure of a gas having volume 100 lits. originally occupying 75 dm3 at 700 mm. pressure.
Assuming temperature remaining constant calculate the pressure of the gas in the following:
The pressure of a gas having volume 1500 cm3 originally occupying 750 cc. at 5 ats. pressure.
According to Boyle’s law, the shape of the graph between pressure and reciprocal of volume is _______.
State Boyle’s law
