Advertisements
Advertisements
Question
Correct the following statement:
The volume of a gas is inversely proportional to its pressure at a constant temperature.
Solution
The volume of a given mass of a dry gas is inversely proportional to its pressure at a constant temperature.
APPEARS IN
RELATED QUESTIONS
State the law which is represented by the following graph:
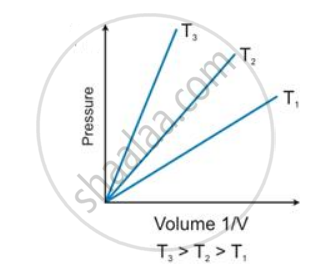
Give reasons for the following:
It is necessary to specify the pressure and temperature of gas while stating its volume.
561 dm3 of a gas at STP is filled in a 748 dm3 container. If the temperature is constant, calculate the percentage change in pressure required.
A certain amount of a gas occupies a volume of 0.4 litre at 17°C. To what temperature should it be heated so that its volume gets (a) doubled, (b) reduced to half, pressure remaining constant?
Calculate the volume of dry air at STP that occupies 28 cm3 at 14°C and 750 mmHg pressure when saturated with water vapour. The vapour pressure of water at 14°C is 12 mmHg.
Assuming temperature remaining constant calculate the pressure of the gas in the following:
The pressure of a gas having volume 100 lits. originally occupying 75 dm3 at 700 mm. pressure.
Assuming temperature remaining constant calculate the pressure of the gas in the following:
The pressure of a gas having volume 1800 ml. originally occupying 300 ml. at 6 atms. pressure.
Assuming temperature remaining constant calculate the pressure of the gas in the following:
The pressure of a gas having volume 1500 cm3 originally occupying 750 cc. at 5 ats. pressure.
Fill in the blank with the correct word, from the words in option:
1 dm3 of a gas is equal to _______.
State-the law of volume
