Advertisements
Advertisements
प्रश्न
Fill in the blank with the correct word, from the words in option:
1 dm3 of a gas is equal to _______.
विकल्प
1 litre
100 ml.
100 cc.
उत्तर
1 dm3 of a gas is equal to 1 litre.
APPEARS IN
संबंधित प्रश्न
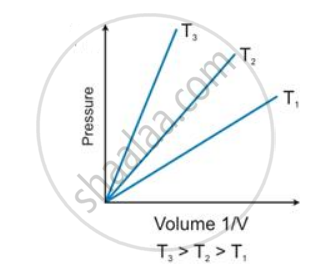
State the law which is represented by the following graph:
A steel cylinder of internal volume 20 litres is filled with hydrogen at 29 atmospheric pressure. If hydrogen is used to fill a balloon at 1.25 atmospheric pressure at the same temperature, what volume will the gas occupy?
A certain amount of a gas occupies a volume of 0.4 litre at 17°C. To what temperature should it be heated so that its volume gets (a) doubled, (b) reduced to half, pressure remaining constant?
At 0°C and 760 mmHg pressure, a gas occupies a volume of 100 cm3. Kelvin temperature of the gas is increased by one-fifth and the pressure is increased one and a half times. Calculate the final volume of the gas.
Calculate the volume of dry air at STP that occupies 28 cm3 at 14°C and 750 mmHg pressure when saturated with water vapour. The vapour pressure of water at 14°C is 12 mmHg.
Give its
(i) mathematical expression
(ii) graphical representation and
(iii) significance.
A given mass of a gas occupied 143 cm3 at 27° C and 700 mm Hg pressure. What will be its volume at 300 K and 280 mm Hg pressure?
Calculate the volume of a gas ‘A’ at s.t.p., if at 37°C and 775 mm of mercury pressure, it occupies a volume of `9 1/2` litres.
Assuming temperature remaining constant calculate the pressure of the gas in the following:
The pressure of a gas having volume 1500 cm3 originally occupying 750 cc. at 5 ats. pressure.
Calculate the coefficient of cubical expansion of a zinc bar. Whose volume is increased 0.25 m3 from 0.3 m3 due to the change in its temperature of 50K?
