Advertisements
Advertisements
प्रश्न
Explain with a diagram the process used to – separate the following substance from the given mixture.
Potassium nitrate from a mixture of – potassium nitrate & potassium chlorate.
उत्तर
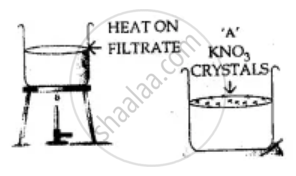
Potassium nitrate KNO3 is more-soluble than potassium chlorate KClO3.
On heating to get the saturated solution and on cooling the saturated solution less soluble (KClO3) crystallise out. More soluble KNO3 is filtered out from the hot saturated solution and is recrystallised from hot water and dried.

APPEARS IN
संबंधित प्रश्न
Why is iron sulphide a compound ?
Choose the most appropriate answer
Which of the following property is not shown by compounds?
Why is filtration a better technique than sedimentation and decantation ?
Name the following:
The heavier liquid component from mercury and water.
Explain with diagram the process used to – separate the following substance from the given mixtures - Different dyes – in their liquid constituent ink.
Visit a nearby paddy field and rice mill and note down the different separating techniques used there. Is technology replacing some traditional practices?
Name the components in each of the following mixtures.
- Ice cream
- Lemonade
- Air
- Soil
How will you separate sand and water from their mixture?
Match the following
| Column A | Column B | ||
| 1. | Separation of salt from saltwater | a. | Magnetic separation |
| 2. | Separation of tea leaves from tea extract | b. | Evaporation |
| 3. | Separation of sand and iron filings | c. | Churning |
| 4. | Separation of butter from curds | d. | Filtration |
A mixture of iron nails, salt, oil and water is provided to you. Give stepwise methods to separate each component from this mixture.
