Advertisements
Advertisements
प्रश्न
Explain with a diagram the process used to – separate the following substance from the given mixture.
Potassium nitrate from a mixture of – potassium nitrate & potassium chlorate.
उत्तर
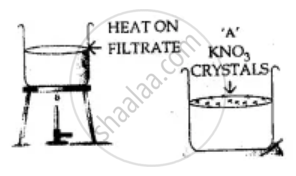
Potassium nitrate KNO3 is more-soluble than potassium chlorate KClO3.
On heating to get the saturated solution and on cooling the saturated solution less soluble (KClO3) crystallise out. More soluble KNO3 is filtered out from the hot saturated solution and is recrystallised from hot water and dried.

APPEARS IN
संबंधित प्रश्न
Differentiate between:
pure and impure substances
Write true or false
Milk is an emulsion.
Suggest a suitable technique to separate the constituents of the following mixtures. Also give the reason for selecting the particular method.
Chalk powder from water
Suggest a suitable technique to separate the constituents of the following mixtures. Also give the reason for selecting the particular method.
Calcium carbonate and sodium chloride
Match the ideal method of separation of components in a mixture in List I with the appropriate process in List II.
| List I | List II |
| 1. Sand from a mixture of sand and water | A: Separating funnel |
| 2. kerosene from a mixture of kerosene water | B: Sublimation |
| 3. Alcohol from a mixture of methyl alcohol and water | C: Filtration |
| 4. Naphthalene from a mixture of naphthalene and lead chloride | D: Distillation |
| 5. Pure water from impure water | E: Fractional distillation |
State any one method – to separate the following mixtures – Two solid mixtures one of which – directly changes into vapour on heating.
State any one method – to separate the following mixtures – Two solid mixtures one of which – dissolves in a – particular solvent and other does not
Very Short Answer Type Question:
What types of material can we separate by using handpicking?
Name the apparatus that you will use to separate the components of mixtures containing two,
- miscible liquids,
- immiscible liquids
Match the following
| Column A | Column B | ||
| 1. | Separation of salt from saltwater | a. | Magnetic separation |
| 2. | Separation of tea leaves from tea extract | b. | Evaporation |
| 3. | Separation of sand and iron filings | c. | Churning |
| 4. | Separation of butter from curds | d. | Filtration |
