Advertisements
Advertisements
प्रश्न
Explain with the help of a balanced equation, the brown ring test for nitric acid.
उत्तर
Procedure:
- Add freshly prepared saturated solution of iron (II) sulphate to the aq. solution of nitric acid.
- Now add conc. sulphuric acid carefully from the sides of the test tube so that it does not fall dropwise in the test tube.
- Cool the test tube in water.
- A brown ring appears at the junction of the two liquids.
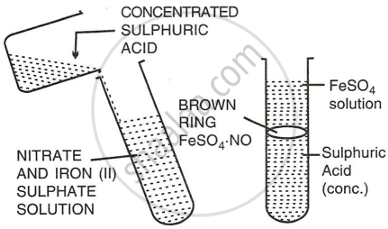
Reaction:
\[\ce{6FeSO4 + 3H2SO4 + 2HNO3 -> 3Fe2[SO4]2 + 4H2O + 2NO}\]
\[\ce{FeSO4 + NO -> FeSO4 * NO}\]
APPEARS IN
संबंधित प्रश्न
Explain the following:
An all-glass apparatus is used in the laboratory preparation of nitric acid.
Describe the Ostwald's process for the manufacture of nitric acid with labeled diagram.
Give three equations of the reaction to prove that nitric acid is an acid.
Complete and balance the following equation :
HNO3 + FeSO4 + H2SO4 → ______________
Fill in the blank with appropriate word/words.
Aqua regia is a mixture of _______ and________
Name the compounds required for the laboratory preparation of nitric acid.
The figure given below illustrates the apparatus used in the laboratory preparation of nitric acid:
(i) Name A (a liquid), B( a Solid) and C(a liquid).
(ii) write the balanced chemical equation to show how nitric acid undergoes decomposition.
(iii) Write the balanced chemical equation for the reaction in which copper is oxidized by concentrated nitric acid.
State, what is observe when nitric acid is kept in a reagent bottle for a long time.
Write a balanced equation for following :
Laboratory preparation nitric acid.
Explain why nitric acid is kept in a reagent bottle for a long time.
