Advertisements
Advertisements
Question
Explain with the help of a balanced equation, the brown ring test for nitric acid.
Solution
Procedure:
- Add freshly prepared saturated solution of iron (II) sulphate to the aq. solution of nitric acid.
- Now add conc. sulphuric acid carefully from the sides of the test tube so that it does not fall dropwise in the test tube.
- Cool the test tube in water.
- A brown ring appears at the junction of the two liquids.
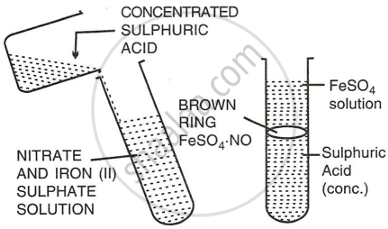
Reaction:
\[\ce{6FeSO4 + 3H2SO4 + 2HNO3 -> 3Fe2[SO4]2 + 4H2O + 2NO}\]
\[\ce{FeSO4 + NO -> FeSO4 * NO}\]
APPEARS IN
RELATED QUESTIONS
Explain the following:
An all-glass apparatus is used in the laboratory preparation of nitric acid.
Ammonia is used in the Ostwald process.
What is the ratio of ammonia and air taken in this process?
Name the process used for the manufacture of nitric acid.
Give three equations of the reaction to prove that nitric acid is an acid.
Write balanced equation and name the product formed when :
Sodium hydrogen carbonate is added to nitric acid.
Write balanced equation and name the product formed when :
Zinc reacts with dilute nitric acid.
Fill in the blank with appropriate word/words.
Ammonal is a mixture of ________ and __________
Give the name of a soluble lead salt and write the eqution for the action of heat on this salt. Write the equation for the preparation of nitric acid from potassium nitrate.
Name the gas evolved:
The gas produced on heating sodium nitrate.
Give one test to distinguish between the following pair of chemicals.
Zinc nitrate solution and calcium nitrate solution.
