Advertisements
Advertisements
प्रश्न
Explain with the help of a balanced equation, the brown ring test for nitric acid.
उत्तर
Procedure:
- Add freshly prepared saturated solution of iron (II) sulphate to the aq. solution of nitric acid.
- Now add conc. sulphuric acid carefully from the sides of the test tube so that it does not fall dropwise in the test tube.
- Cool the test tube in water.
- A brown ring appears at the junction of the two liquids.
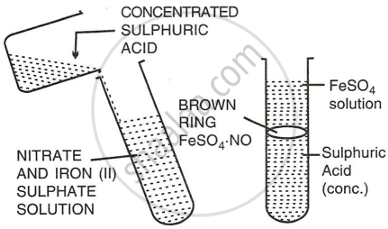
Reaction:
\[\ce{6FeSO4 + 3H2SO4 + 2HNO3 -> 3Fe2[SO4]2 + 4H2O + 2NO}\]
\[\ce{FeSO4 + NO -> FeSO4 * NO}\]
APPEARS IN
संबंधित प्रश्न
Name the products formed when carbon and conc. nitric acid is heated.
How will you prepare the following from nitric acid?
Lead nitrate
Name a metal nitrate which on heating is changed into metal oxide.
What is meant by the following term ?
Decrepitation
How is nitric acid prepared in laboratory?
Name the process used for the manufacture of nitric acid.
Fill in the blank with appropriate word/words.
3 Cu + 8 HNO3 (dilute) → _______ + 4H2O + _________
Complete and balance the following equation:
Cu + HNO3 → NO2 + ________+ _________
Explain why nitric acid is kept in a reagent bottle for a long time.
A dilute acid B does not normally give hydrogen when reacted with metals but does give a gas when reacts with copper. Identify B. Write the equation with copper.
