Advertisements
Advertisements
प्रश्न
Find out the most stable species from the following. Justify.
\[\ce{\overset{+}{C}H3, \overset{+}{C}H2Cl, \overset{+}{C}Cl3}\]
उत्तर
The most stable species from the given species is `overset(+)("CH"_3)`
This because it does not contain Cl atom, which exhibits an electron-withdrawing inductive effect. Carbocations are destabilized by -I inductive (electron-withdrawing) effect. When more number of –I groups are attached to the positively charged carbon atom, the positive charge on the carbon atom increases further, thus destabilizing the species. Hence, the species with no –I groups will be most stable.
APPEARS IN
संबंधित प्रश्न
Find out the most stable species from the following. Justify.
`bar"C""H"_3, bar"C""H"_2"Br", bar"C""Br"_3`
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH_3 - CH_2 - \overset{⊕}{C}H-CH_2 - CH_3}\]
Draw a resonance structure of the following:
Phenol
Draw a resonance structure of the following:
Benzaldehyde
Distinguish between Electrophile and nucleophile.
Choose the correct option.
The geometry of a carbocation is ______.
Predict the order of reactivity of the following compounds by SNl reaction mechanism.
\[\ce{\underset{\text{(I)}}{C6H5CH(C6H5)Cl}}\]
\[\ce{\underset{\text{(II)}}{C6H5CH2Cl}}\]
\[\ce{\underset{\text{(III)}}{C6H5C(CH3)(C6H5)Cl}}\]
Which of the following is NOT an electrophile?
Which of the following is TRUE for homolytic fission?
Identify the reagent used in the following reaction:
\[\ce{CH3 - CH2 - Br ->[?] CH3 - CH2 - OH}\]
The best reagent for the following conversion is:
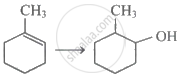
Which of the following alkyl groups shows least positive inductive effect?
Resonance is NOT exhibited by ____________.
Which of the following statements is not correct?
Which element among the following does form pπ - pπ multiple bonds?
Identify the group that exerts electron withdrawing resonance effect.
Identify the functional group that has an electron-donating inductive effect.
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α-carbon in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen in each.
\[\ce{CH3 - CH2 - \overset{\oplus}{C}H -CH2 - CH2 }\]
Identify the α-carbon in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen in each.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α - carbon in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{\oplus}{C}H -CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
