Advertisements
Advertisements
प्रश्न
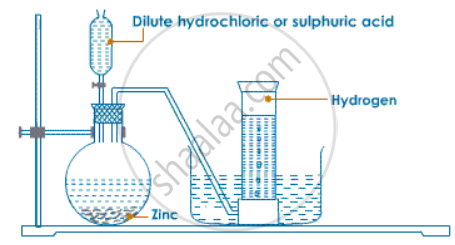
For laboratory preparation of hydrogen, give the following:
fully-labeled diagram
उत्तर
APPEARS IN
संबंधित प्रश्न
Fill in the blank
Hydrogen is ..................... than air.
Which test should be made before collecting hydrogen in a gas jar?
Why hot concentrated sulphuric acid is not used in the preparation of hydrogen?
Hydrogen may be prepared in the laboratory by the action of a metal on an acid.
Which of the metals copper, zinc, magnesium or sodium would be the most suitable?
Hydrogen may be prepared in the laboratory by the action of a metal on an acid.
Which of the acids dilute sulphuric, concentrated sulphuric, dilute nitric acid and concentrated nitric acid would you choose? Explain why you would not use the acids you reject.
Give reason:
A candle brought near the mouth of a jar containing hydrogen gas starts burning but is extinguished when pushed inside the jar.
Select the correct options for the following statement:
The product formed on combustion of hydrogen in air is ___.
Complete and balance the equation:
[General method]
Reactions of metals with dilute acids
Aluminium - 2Al + 3H2SO4 → ______+ _______ [g]
Give a balanced equation for obtaining hydrogen from?
Steam using a trivalent metal
Select the correct answer to the reactant added, to give the product in the preparation of hydrogen gas.
\[\ce{MgO + H2}\]
