Advertisements
Advertisements
प्रश्न
Give a reason for the following:
Mn+2 compounds are more stable than Fe+2 compounds.
उत्तर
Mn2+ = 1s2 2s2 2p6 3s2 3p6 3d5 (Half-filles d-orbital)
Fe2+ = 1s2 2s2 2p6 3s2 3p6 3d6
Mn2+ molecules are more stable because of their half-filled d-orbitals. Fe2+ complexes are less stable because they have six electrons in the 3d-orbital. Hence, they loose one electron to get stable 3d5 configuration.
APPEARS IN
संबंधित प्रश्न
Draw structure of Chlorine pentafluoride
Fluorine is a stronger oxidising agent than chlorine. Why?
Why are halogens coloured?
Complete the following equations:
KMnO4 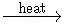
Cl2 acts as a bleaching agent.
Fluorine differs from the rest of the halogens in some of its properties. This is due to ____________.
Which of the following pairs of ions are isoelectronic and isostructural?
Which of the following statements are true?
(i) Only type of interactions between particles of noble gases are due to weak dispersion forces.
(ii) Ionisation enthalpy of molecular oxygen is very close to that of xenon.
(iii) Hydrolysis of XeF6 is a redox reaction.
(iv) Xenon fluorides are not reactive.
Give reason to explain why ClF3 exists but FCl3 does not exist.
Statement I: Acid strength increases in the order given as HF << HCl << HBr << HI.
Statement II: As the size of the elements F, Cl, Br, and I increase down the group, the bond strength of HF, HCl, HBr, and HI decreases and so the acid strength increases.
In the light of the above statements, choose the correct answer from the options given below.
