Advertisements
Advertisements
प्रश्न
Give an example for a complex of the type [Ma2b2c2] where a, b, c are monodentate ligands and give the possible isomers.
उत्तर
The octahedral complexes of [Ma2b2c2] type can exist in five geometrical isomers.
The five geometrical isomers for the complex ion [PtCl2(NH3)2(py)2]2+are shown below.
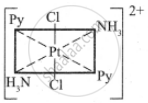
(I)
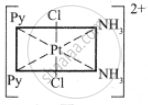
(II)
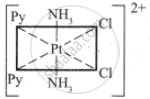
(III)
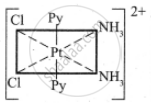
(IV)
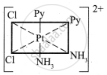
(V)
APPEARS IN
संबंधित प्रश्न
The sum of primary valence and secondary valence of the metal M in the complex [M(en)2(Ox)]Cl is ____________.
As per IUPAC guidelines, the name of the complex [Co(en)2(ONO)Cl]Cl is
Crystal field stabilization energy for high spin d5 octahedral complex is ____________.
Choose the correct statement.
Arrange the following in order of increasing molar conductivity.
i) \[\ce{Mg[Cr(NH3) (Cl)5]}\]
ii) \[\ce{[Cr(NH3)5 Cl]3 [CoF6]2}\]
iii) \[\ce{[Cr(NH3)3 Cl3]}\]
Give one test to differentiate \[\ce{[Co(NH3)5Cl]SO4}\] and \[\ce{[Co(NH3)5SO4]Cl}\].
Why tetrahedral complexes do not exhibit geometrical isomerism.
What is crystal field splitting energy?
What is crystal field stabilization energy (CFSE)?
A solution of \[\ce{[Ni(H2O)6]^2+}\] is green, whereas a solution of \[\ce{[Ni(CN)4]^2-}\] is colorless – Explain.
